Part 1: When to Oxygen Clean

FIRES IN COMPRESSED OXYGEN EQUIPMENT are rare, even though we hear about life-threatening accidents — a rebreather fire, an emergency oxygen regulator accident, or a gas blending system fire.
Several key questions arise: Why do situations with high oxygen levels and dirty hands or workbenches not always result in a fire? What is sufficient oxygen in a breathing gas to elevate the fire risk?
The answer to the first question lies in the statistical term probabilistic. Sometimes materials in 100 percent oxygen at 2,000 psi and heated to 400°F spontaneously ignite. Sometimes they don’t. If there is a reason for this, it is likely beyond even the most qualified scientists to explain fully. But we must err on the side of caution. No oxygen fire is acceptable, and significant injuries or fatalities are often the outcomes.
The number of oxygen molecules in an air-filled scuba cylinder at 3,000 psi is 207 times more than what we find in the atmosphere. There are other factors, but even a charged scuba cylinder could be considered oxygen enriched.
Fires in our air systems are extremely rare. Setting aside the ignition sources and flammable materials, let us focus on situations where the amount of oxygen increases the risk of fire.
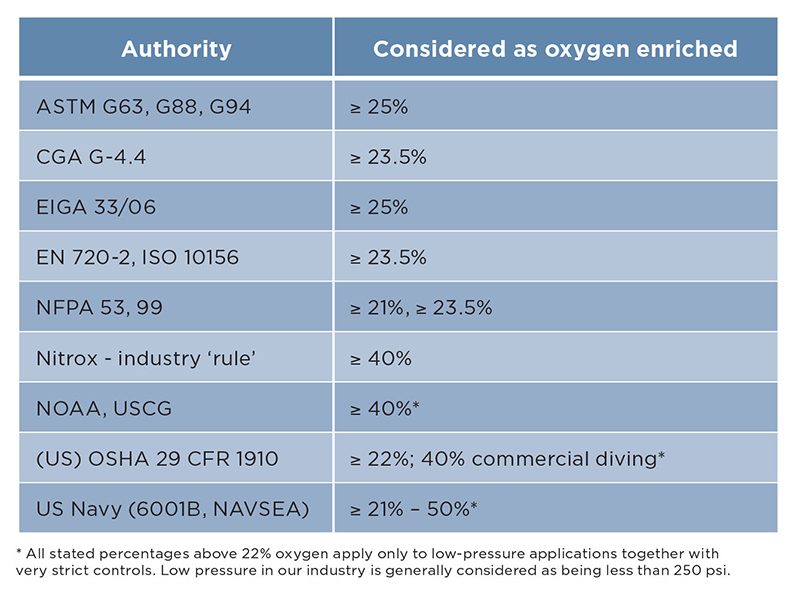
Many codes, standards, specifications, guidelines, and industry practices provide differing definitions of oxygen-enriched breathing gas. We hear statements that 40 percent nitrox does not require oxygen-clean equipment. How do we know the acceptable, safe level with all this conflicting information?
Table 1 summarizes references stating when gas is oxygen enriched. Realistically, we could draw the line at no more than 25 percent, but it is important to ask where the level was tested, the intended maximum pressure, and how oxygen gets above 21 percent.
Navy research in the 1960s and ’70s included studies of fires with varying oxygen-enrichment levels, breathing-gas mixtures, and environmental pressures. The results showed that as pressure increases, the flame rate of materials increases rapidly with increasing oxygen concentrations. Above 25 percent, the burn rate rises rapidly. At 40 percent oxygen and 90 psi, the burn rate is 50 percent higher than the 21 percent oxygen in normal air. At higher pressures, 40 percent quickly becomes a fireball.
For lower-pressure applications, such as in commercial diving umbilicals, the oxygen levels could be higher than 25 percent. At the 3,000 psi of a scuba cylinder, all bets are off.
The American Society for Testing and Materials standard for gear material selection and design provides the most defendable answer: For use at 25 percent and above, all high-pressure scuba gear should be cleaned for oxygen use.
In the next issue of Alert Diver, Part 2 will cover the criteria for oxygen cleaning.
Alert Diver — Q2 2022