Divers first learn about the malady known as decompression sickness (DCS) during open-water training. DCS occurs when our body’s tissues absorb inert gas (such as nitrogen) during a dive and the gas comes out of solution during ascent, forming bubbles. These bubbles can cause tissue injury, blocked blood vessels and a cascade of physiological responses. DCS symptoms can include skin rash, joint pain, malaise (general discomfort), dizziness, muscle weakness, shortness of breath, paralysis and unconsciousness.
Unfortunately, with the exception of blood vessel blockage, the exact causes of these symptoms are still poorly understood. The prevailing idea is that DCS is caused by activation of the inflammation, coagulation and/or systemic immune responses by the physical effects of the bubbles; however, research is still under way to determine to what degree these responses are involved in DCS.
Biomarkers
Developing new ways to study the causes and degrees of stress the body experiences during decompression will benefit not only divers but also high-altitude pilots and astronauts. A blood-based indicator of decompression stress (DS) could be used as a biological marker, or biomarker, in human decompression studies, providing a clearer picture of the molecular mechanisms involved in DCS. A DS-related biomarker could also be used as a starting point for developing a test to diagnose DCS in real time.
The Massachusetts General Hospital’s Center for Biomarkers in Imaging defines biomarkers as “anatomic, physiologic, biochemical or molecular parameters associated with the presence and severity of specific disease states.” The National Institutes of Health defines biomarkers as “a characteristic that is objectively measured and evaluated as an indicator of normal biologic processes, pathogenic processes or pharmacologic responses to therapeutic interventions.” Molecular biomarkers are chemical or molecular structures that indicate disease, injury or stressors in the body. Biomarkers include anything from a single molecule or macromolecule to a complex signature of molecular changes based on a particular pathology. Biomarkers can be useful in diagnosing clinical conditions, predicting clinical outcomes or risks and providing objective measurements of ongoing diseases or injury processes.
Diving research studies have investigated a selection of possible biomarkers of DS and DCS. For decades researchers have observed blood biochemical parameters (such as enzyme levels and metabolic products) in decompression studies; however, research hasn’t established any direct correlation between these parameters and DCS incidence or severity.
Still, researchers continue to look into markers of other medical conditions as candidates. For example, D-dimer, which is a small protein fragment that results from the degradation of blood clots and serves as a marker of neurological injury, has been measured at increased levels in divers with DCS who exhibited neurological deficits. S100ß, another marker of neurological injury, has been shown to increase in animal subjects following rapid decompression. Researchers have also found that levels of brain natriuretic peptide in the plasma start to increase in divers after one hour underwater at 33 feet and remain elevated for four hours after surfacing.
As with blood biochemical parameters, however, no neurological marker has been established as a potential marker of DS or DCS. Previous studies have also shown that creatine phosphokinase, an indicator of tissue damage in disease and injury, elevates significantly in subjects diagnosed with DCS and correlates with the presence of arterial bubbles. Unfortunately, none of these candidates have been established as a reliable biomarker of DS or DCS.
Background
To search for candidate biomarkers in blood, my research team at Duke University uses a technique called expression microarray, which measures the expression level of every gene present in our cells. Genes contain the basic information needed to assemble molecules called proteins, which are the building blocks of our bodies. Genes also regulate how and where different proteins are made and used in our bodies. When the body’s tissues are exposed to stressors (such as heat, infection, injury or gas bubbles), certain genes will be expressed (“turned on”) or repressed (“turned off”) to initiate a response to that stressor, which amounts to production of more or less of that gene’s corresponding protein.
Genetic information in cells flows from DNA to RNA to proteins. Microarray technology is able to measure the amount of RNA from every gene in a cell, and this provides information about the relative expression level of each gene in a sample of blood or tissue. We conducted a study to investigate the relative expression level of every gene in circulating blood cells to provide insight into new gene marker candidates and molecular pathways involved in DS.
Methods
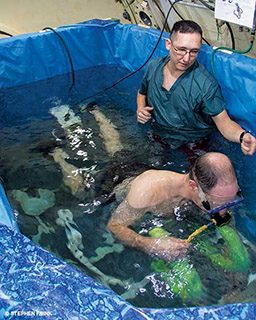
Using expression microarray we measured the expression levels of more than 14,000 genes from human venous blood samples collected one hour before and two hours after experimental dives. The subjects were submerged and exercising in a hyperbaric chamber, which allowed the study team to precisely control the experimental dive profile to minimize potential sources of variance. We compared predive and postdive samples to identify significant genes and the corresponding biochemical pathways in response to DS. None of the subjects experienced symptoms of DCS.
The subjects breathed hyperbaric oxygen during decompression. Because changes in gene expression following the experimental dives could have been related to breathing high partial pressures of oxygen, we conducted a separate experiment to analyze blood gene expression one hour before and two hours after breathing hyperbaric oxygen. Similarly, since the subjects exercised during the bottom phase of the dive, we assessed changes in blood gene expression following exercise of similar exertion and duration in a separate experiment.
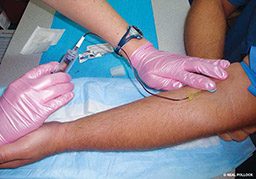
Finally, we addressed the effect of time of day on gene expression by drawing samples at approximately 7 a.m. and 5 p.m. for the experimental dive series. We drew blood from a separate experimental group with no other experimental exposures at 7 a.m. and 5 p.m. to help us identify those genes that are expressed at different levels solely based on time of day. We processed the blood samples to extract the RNA from the cells and then processed the RNA to measure the relative expression levels of more than 14,000 genes using expression microarray.
Summary
Statistical analyses revealed that more than 700 genes (out of the more than 14,000 we measured) were expressed at levels that were significantly higher or lower following the experimental dives compared to predive baseline measurements.
In the analysis of the hyperoxic exposure control group, approximately 200 genes were expressed compared to baseline levels, including genes associated with cell communication and cell adhesion. The analysis of blood samples collected to account for time of day demonstrated approximately 30 genes that were differentially expressed between 7 a.m. and 5 p.m. With respect to exercise effect, there were no significant changes in gene expression two hours after exercise compared to baseline levels.
After filtering out genes that were turned on or off due to time of day or hyperoxic exposure, the study still revealed approximately 700 genes that were expressed or repressed at significant levels following experimental diving exposures. These results demonstrate that a significant number of peripheral blood genes are differentially regulated in response to diving — independent of hyperoxic exposure, exercise or time of day. A significant number of these genes are associated with innate immune and inflammatory responses.
Inspect
In this study we identified a set of key genes and gene-level pathways associated with decompression stress as a first step in identifying potential biomarker candidates of DS and DCS. Understanding the mechanism of an injury, response or disease process is useful, if not necessary, for identifying possible biomarkers of that specific condition.
Several pathways have been suggested as underlying mechanisms of DS and DCS in humans. Increased clotting activity and evidence of coagulation have been demonstrated in cases of DCS and in asymptomatic divers following hyperbaric exposures. Platelet activation has also been detected after asymptomatic scuba dives. Studies have demonstrated elevation of inflammatory and immune markers following asymptomatic dives and in cases of DCS.
Previous work by Bruce Cameron and colleagues investigated the expression of specific immunoinflammatory genes in military divers following deep air dives. Similar to the results in our study, a number of immunoinflammatory genes were found to be differentially expressed postdive. A follow-up study by the same group assessed peripheral blood collected pre- and postdive to detect significant differences in gene expression following a series of mixed-gas dives. Differential expression of immunoinflammatory genes was observed postdive in all of the profiles.
A recent microarray-based study by Ingrid Eftedal and colleagues collected blood samples before and immediately after the first and last dives of a three-day series of daily open-water dives on compressed air. These researchers found significant changes in immune and inflammatory cell gene expression after diving. Antioxidant genes were also upregulated. The authors concluded that extensive diving may cause persistent change in pathways controlling cell death, inflammation and innate immune responses.
Jan Krog and colleagues recently suggested that immune-related markers would be ideal candidates as DS biomarkers. Their conclusion was based on the increased number of circulating natural killer (NK) cells and the increase in NK cell toxicity they observed in military divers following decompression from deep saturation. Although activation of these pathways has been demonstrated following decompression exposures or development of DCS, no biomarkers of DS or predictors of DCS have been established.
Our study investigated the changes in expression of thousands of genes in human blood samples following controlled diving exposures, and the study design controlled for other potential sources of biological variance (hyperoxia, time of day and exercise). The development and validation of a DS marker or panel of markers is the first step in identifying a test for use in human experimental decompression studies. Such a test could also provide the basis for a diagnostic test of DCS that could be used in the treatment setting.
Ongoing studies are investigating multigene, single gene and related protein candidates associated with these pathways as possible blood markers of DS. Developing an improved understanding of the pathways associated with decompression exposures will not only establish optimal candidates for DS and DCS diagnostic markers, but it will also provide insight into the molecular and tissue-level mechanisms underlying the body’s response to bubble formation, presence, damage and other potential stressors associated with decompression.
© Alert Diver — Q2 Spring 2015